A02 Ito and Matsuoka’s paper on
Happy to share A02 Ito and Matsuoka’s latest research on L-region selective APEX reaction for nanographene synthesis!! This is a first demonstration of L-region (C1,C2-position of naphthalene) APEX, and also part of the themed collection: Celebrating 200 Years of Benzene!! The paper was also highlighted as an Inside Cover Art. Congratulations!!
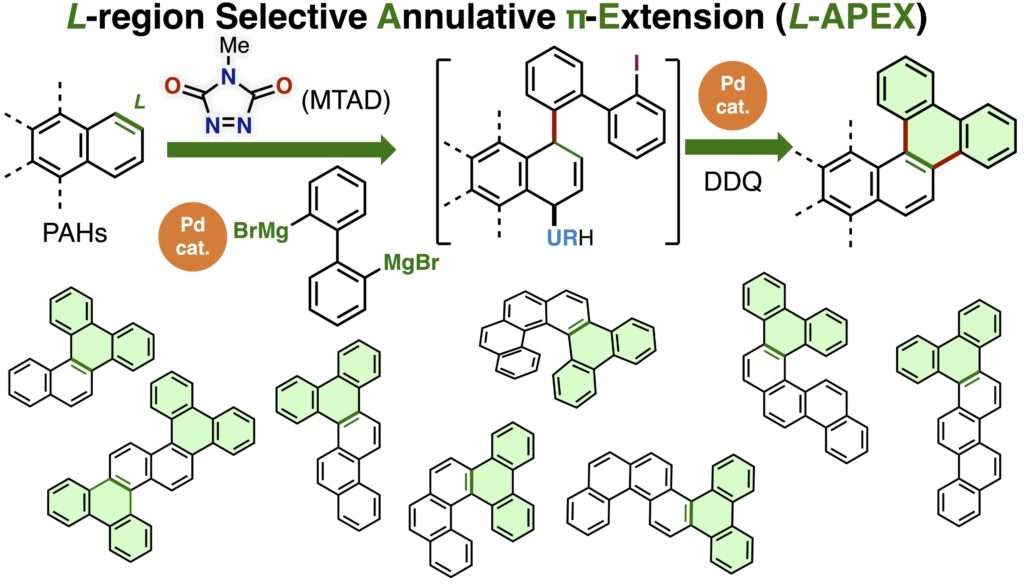
L-Region-selective annulative π-extension through dearomative activation of polycyclic aromatic hydrocarbons
Kanami Nakata, Wataru Matsuoka, Hideto Ito* and Kenichiro Itami*
Chem. Sci. 2026, 17, 3998–4003. DOI: 10.1039/D5SC09309K
Selected as an Inside Front Cover Art.
Part of themed collections “Celebrating 200 Years Benzene”
Annulative π-extension (APEX) reaction is a useful aromatic ring-fusion method for the synthesis of large polycyclic aromatic hydrocarbons (PAHs) from unfunctionalized small PAHs. While APEX reactions in the K-, M-, and bay-regions of PAHs have been developed, L-region selective APEX is yet to be achieved. Herein, we report a stepwise L-region selective APEX of unfunctionalized PAHs by dearomative activation with N-methyltriazoline dione, followed by Pd-catalyzed annulation with aryl Grignard reagents. Various difficult-to-synthesize core-expanded PAHs can be synthesized by L-APEX from unfunctionalized naphthalene, phenanthrene, chrysene, and [4]helicene.